New ways to achieve comparable and reproducible findings
Vehicle fuel cells contain many non-metals, particularly polymers, for several different purposes. While seals are made from elastomers, Type IV containers are lined with thermoplastics, the same materials that are now also increasingly used to make hardware parts, such as gaskets. The standards and guidelines relevant to the industry require a series of tests to ensure that the chosen polymers are suitable for a given application.
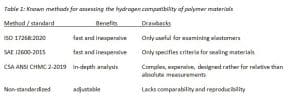
The ozone aging and oxygen deterioration tests required by EU directives 79/2009 and 406/2010, and similarly UN R134, are comparatively well specified and reference tried-and-proven, standardized test methods. Although hydrogen compatibility tests are also part of the regulations, the rules describing the conditions in which these examinations are to be performed are rather vague, relatively imprecise or non-existent. The most likely reason for this is that just a few years ago, there were still considerable gaps in standardization, a problem compounded by the lack of hydrogen expertise in the auto industry when the standards were created.
Rules disappoint
In combination with EU directive 406/2010, EC 79/2009 is often cited when issuing a certificate of conformity for vehicles sold on the European market. While the directive lists specific procedures that must be followed when analyzing metallic materials, i.e., ISO 11114-4 examinations of components or pressure cycling tests for Type I, II and III storage tanks, the regulations on non-metallic components consist of one simple sentence: “Hydrogen compatibility shall be demonstrated.”
Additionally, UN GTR 13, or, more specifically, UN R134, make no mention of assessing the compatibility of non-metals, instead referring only to oxygen and ozone deterioration. Despite these missing specifications, proof of hydrogen compatibility is crucial, especially for safety and liability reasons.
The lack of standardized procedures has led to the creation of a variety of methods, and each has its own benefits and drawbacks. Some examples can be found in table 1.
While an optimal test method obviously needs to deliver meaningful, clear and reproducible results, economic viability should also be part of the equation. In many cases, several potentially compatible materials need to be examined at a relatively early stage of development. The findings are then used to select the most promising among them, with a focus on choosing the least expensive material(s).
One quick and easy-to-conduct method is described in SAE J2600. It involves measuring the mass and volume of a sample on a scale or in a liquid bath, followed by a 168-hour static hold in a hydrogen environment at normal operating pressure and room temperature (or at minimal process temperature of mainly around -40 °C). Shortly after the time has elapsed, the container holding the sample is depressurized to ambient to encourage explosive decompression. Subsequently, mass and volume are measured a second time to see whether decompression of the material has occurred, indications being bubbles or ruptures or both, and whether the sample shows signs of increased swelling or shrinkage or a loss in mass.
The method has its drawbacks, however. The associated standard was basically designed to examine only two specific fuel cell system components, the receptacle and the fueling nozzle, which means that it may have limited applicability elsewhere. In addition, SAE analyses make sense only in the context of elastomer (valve) materials. For example, the 25-percent swelling threshold listed in the standard is not relevant to thermoplastics. Consequently, thermoplastics often undergo alternative, individually designed tests. These, however, have their own disadvantages:
• Some offer limited reproducibility.
• Some also require complicated sample geometries, increasing production costs.
• Their non-standardized procedures lack formal recognition.
• There is a risk that the interpretation of procedures and data will vary among departments or organizations. As a result, the findings may only apply to new products.
It is thus in the interest of several groups, including automakers, suppliers, test institutes and laboratories, to find a solution that has none of the disadvantages mentioned above and delivers both valid and reproducible results while being inexpensive and fast.
Standardizing hydrogen compatibility tests
The authors of this article are offering a new, easy method to prove hydrogen compatibility for polymers. This method can be performed on both elastomers and thermoplastics and meets current regulatory requirements.
…
Enjoy reading more in the latest edition of the H2-International October 2020
Sources and references available on request.
Author:
Martin Sekura
TesTneT Engineering GmbH
sekura@h2-test.net
Dr. Stefan Behrning
TÜV Saarland Automobil GmbH























0 Comments