The growing importance of nickel, tin and copper
New times call for new ideas – and new materials. A global increase in electrification calls for new chemical products. Petroleum catalysts are a major component of the fossil fuel era and must be replaced. The current battle for rare natural resources and chemicals makes it all the more important to invest in research on inexpensive, readily available alternatives. Evidence points to nickel, tin and copper, and their undiscovered properties, as cheaper options. Good news for the electrolyzer and fuel cell industries.
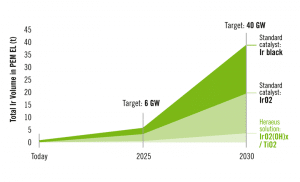
Professor Peter Strasser, leader of TU Berlin’s electrochemical energy, catalyst and materials science group, has been researching nickel and iron oxide catalysts used in alkaline exchange membrane water electrolyzers, or AEMWEs. He aims to find an alternative to iridium catalysts in proton exchange membrane water electrolysis, PEMWE. Strasser explained: “PEMWE catalysts are made from iridium, an expensive, extremely rare precious metal covering the anode, where oxygen is extracted from water. To achieve the 2030 capacity targets in the German government’s hydrogen strategy, we would need the world’s entire yearly iridium supply to meet just a small percentage of Germany’s energy demand.” (see also H2-international, October 2020)
Iridium production is currently around 8 tons annually. PEM electrolyzers need about 1 to 2 grams per kilowatt output. The strategy’s 40-gigawatt electrolyzer target by 2030 would require 20 to 40 tons of iridium a year if PEM devices provided half the capacity.
In May 2020, TU Berlin researchers published an article in Nature Communications, describing new findings and the molecular dynamics of Ni-Fe catalysts. The team compared experimental data with a new theoretical catalyst model developed in partnership with Purdue University in the United States – a model that fully explains and quantifies the results of TU Berlin’s joint experiments with Fritz Haber Institute in Berlin. Strasser said these and similar catalyst research breakthroughs will make a case for water-splitting in “clean, efficient and inexpensive hydrogen production.”
Hanau-based Heraeus Group is carrying out similar research. In late September 2020, the German business introduced a new catalyst not only with 50 to 90 percent less iridium but also trebling catalytic performance, according to Heraeus. A far less expensive, far more efficient PEM electrolysis catalyst, the company added.
“Global iridium reserves simply cannot provide sufficient precious metal to supply all electrolyzers necessary for the European Commission’s hydrogen targets. We are very proud to have created a product so low in iridium.”
Christian Gebauer, hydrogen systems director at Heraeus Precious Metals
Copper in PEM fuel cells
Electrolyzers aren’t the only devices needing catalysts. Researchers have also spent a good number of years trying to find substitutes for those in PEM fuel cells. Professor Julia Kunze-Liebhäuser, a physical chemist at Innsbruck University, Austria, has discovered that copper is indeed an option despite the metal’s commonly assumed inertia. She said: “Our own calculations led us to believe that copper simply can’t be a catalyst. And yet, when we conducted our experiment, we detected high electrocatalytic activity.”
… Read more in the latest H2-International e-Journal, Feb. 2021























0 Comments