Improving waste heat recovery
The pyroelectricity phenomenon – crystals converting thermal energy into power – has been known since antiquity. Our ancestors observed that tourmaline thrown into a fire attracted and then repelled ashes. Seventeenth-century scientists began studying the effect in detail, discovering the electric charges are the result of modified crystal structures.
Pyroelectrics are a subgroup of dielectrics, elastic, non-conductive materials that polarize when subjected to an electric field. Such materials include crystalline structures such as lithium niobate and barium titanate, polymers such as polyvinylidene fluoride, and biominerals such as bones.
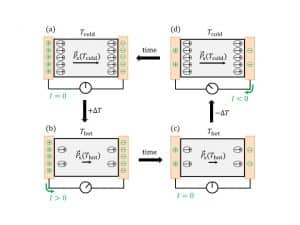
Pyroelectrics are unique in that changes in temperature trigger polarization. Heating or cooling the material shifts positively charged in relationship to negatively charged atoms. These slight shifts in charges add up along basic cells, giving rise to a macroscopically measurable voltage.
Voltage V is influenced by temperature difference , crystal thickness , permittivity , and pyroelectric coefficient . The last is the crystal property that quantifies the pyroelectric effect’s magnitude.
The pyroelectric effect can be used to convert thermal into electrical energy. Thereby, two electrodes are placed on opposite ends of a crystal, which is then subjected to intervals of temperature changes. To maintain an electrical equilibrium, the polarization shift in the material requires charges to be adapted on the electrodes. Surplus charges flow over an outer conductor to the other side of the crystal, where they generate accessible electrical energy.
Charge can be quantified with the help of this second formula, whereby is the cross-section of the crystal.
Pyroelectric catalysis
Obviously, the voltage generated via pyroelectric means can also be used in electrochemical processes. When the metal electrodes are removed, surplus or deficit charges can only flow via chemical reactions, leaving one side of the crystal positive and oxidating, while the other would be negative and reducing (see fig. 2). Redox reactions then convert thermal into chemical energy.
In principle, a variety of chemical reactions is possible, from directly decomposing organic material to inactivating bacteria to water electrolysis for hydrogen production. The hydrogen is separated from the oxygen with a gas diffusion layer and used either to power fuel cells or as an industrial raw material.
Unlike conventional electricity-sourced electrolysis, pyroelectric catalysis can produce power from thermal waste, even low-temperature heat under 100 °C. This waste heat can be retrieved from transportation, food industry or power plant exhaust gases, to name a few. Since global thermal waste comes to several hundred exajoules each year (,according to Forman 2016), these are bright prospects for energy-neutral wastewater treatment and water electrolysis.
… Read more in the latest H2-International e-Journal, Feb. 2021
Authors: Dr. Mateo U. de Vivanco, Dr. Hartmut Stöcker, Prof. Dr. Dirk C. Meyer
All for TU Bergakademie Freiberg, Germany
























0 Comments