At the heart of every PEM fuel cell, there is a membrane electrode assembly. It has a considerable impact on the output and lifetime, as well as the cost, of a stack. It is of such import that it has sparked multiple efforts to research and develop new kinds of materials and manufacturing techniques. A working group made up of scientists from the University of Freiburg, Germany, and the local Hahn-Schickard Institute has been looking into options for the production and microstructural characterization of membrane electrode assemblies, or MEAs for short. The aim is to improve their interfacial properties.
The interfaces between membranes and electrodes are essential to the power output of fuel cells. Yet, most of the time, MEA components are designed individually. It is usually only at a later stage that the hubs of chemical reaction, the electrodes, are added to a thin-film polymer separator during a roll-to-roll process. However, both layers are closely interconnected owing to water management and proton transfer.
As for the water that is produced at an electrode when running a fuel cell, it helps humidify the membrane and has a major influence over the proton conductivity inside the separator. The membrane itself acts as the source of protons to set off chemical reactions at the electrode, so that the highest reaction rate is found in the region between both. Every electrode also contains an ionomer that provides an internal pathway to transfer protons throughout.
The above illustrates how important it is to consider the boundaries between membranes and electrodes when developing PEM fuel cells. It is why the two components must always be treated as an inextricably intertwined unit.
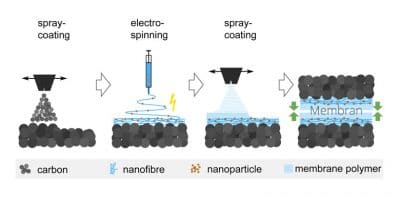
Direct membrane deposition
This year, researchers at the Hahn-Schickard Institute published a study for which they reviewed the findings of about 100 science papers that explore improvements to the interfaces between membranes and electrodes or proton-conducting phases and catalytic centers[1]. The authors concluded that progress had indeed been made. Examples are an increase in ionomer or platinum content toward a membrane and ionomer impregnation on that side of an interface. As reported elsewhere, micropatterned membranes could also allow for substantially larger boundaries[2].
…
read more: H2-international October 2018
Written by: Dr. Matthias Breitwieser, Dr. Severin Vierrath
Both for Hahn-Schickard-Institut, Freiburg, Germany

























0 Comments