The conversion of waste heat into electrical energy could very well make an important contribution to CO2 and GHG emissions reduction and improve energy efficiency. A typical process chain will be based on a thermodynamic cycle, such as organic Rankine, a Stirling engine or a thermoelectric generator. A relatively new method uses pyroelectricity to advance both water electrolysis and power generation. This property of some materials has yet to attract much attention or research. However, considering global efforts to transform the energy market, its importance may grow over time.
Pyroelectricity is a physical property of crystal structures that possess a permanent electric dipole moment. Well-known examples of such materials include barium titanate (BaTiO3), lithium niobate (LiNbO3) and the polymer polyvinylidene fluoride (PVDF). A gradual change in temperature – for example, through adding waste heat – will cause positive and negative charges to migrate to opposite ends of the material and establish an electric potential. This potential can be measured by devices such as infrared thermostats, i.e., contactless temperature probes, or motion sensors, i.e., passive infrared sensors that detect heat emitted by objects and people.
How it works
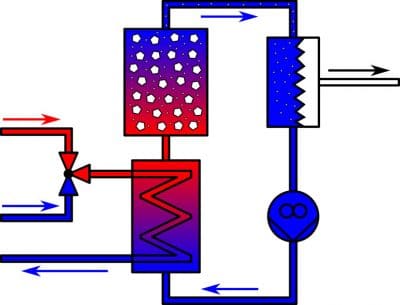
A research team led by Tilmann Leisegang and Hartmut Stöcker from the Institute of Experimental Physics at TU Bergakademie Freiberg has developed a demonstration system that operates based on the above-mentioned phenomenon and can create hydrogen through pyroelectric means. It was first shown to the public at Hannover Messe last year (see fig. 1). The system, which was designed and built over the course of several research projects, uses barium titanate powder and low-temperature waste heat to split water into hydrogen and oxygen. First, externally supplied waste heat is led through a heat exchange into a secondary loop. Then, the water circulating in this loop is directed through a reactor containing the pyroelectric material.
The heating and cooling cycle causes a reaction in the water molecules on the surface of the pyroelectric material, splitting them into oxygen and hydrogen. A subsequent membrane separates the products of the split before they are stored away. This means that the pyroelectric effect makes it possible to convert previously unused waste heat into electrical and chemical energy. In turn, the hydrogen produced by the process can later be utilized in a variety of energy supply scenarios, for example, in hydrocarbon conversion or fuel cell power generation.
…
Alternative to known methods
Methods to convert waste heat into electrical energy have been around for decades. One example is Ormat’s Rankine cycle, which uses organic heat transfer fluids instead of water. The advantage is that heat is transferred at temperatures lower than that of a water-steam cycle, which allows for lower temperatures throughout the entire process.
Compared to two-phase flow units that need to condense and evaporate fluids – from gas to liquid and vice versa – solid-state energy conversion systems have great appeal because of their simple and low-maintenance design. Thermoelectric materials operate based on the Seebeck effect, in which a usable electric potential is created by the generation of a temperature gradient in the solid. One company developing industrial-scale thermoelectric generators for waste heat use is Alphabet Energy.
…
Other uses
Pyroelectric materials can also be employed to convert heat into electricity. The electrical cycles that have been developed to create energy-efficient process chains [2] are akin to thermal ones, but are based on solid connections in which electrons are rearranged to create polarizations and thus energy. The process requires periodic changing of the temperature and polarization, for which several designs are available. The generated electrical energy can be stored or used to generate hydrogen [3].
…
Authors: Dr. Hartmut Stöcker, Professor Dr. Dirk C. Meyer
Both from TU Bergakademie Freiberg, Institut für Experimentelle Physik, Freiberg, Germany


























Your thoughts are welcome regarding how the above process correlates with a projet by French McPhy in Lebanon:
http://hugin.info/160013/R/2121507/808385.pdf
Merely a press release, no in-depth technical information was available.
You should ask directly at TU Bergakademie Freiberg.